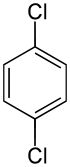
对二氯苯
| 对二氯苯 | |||
|---|---|---|---|
| |||
| IUPAC名 1,4-Dichlorobenzene 1,4-二氯苯 | |||
| 别名 | p-DCB | ||
| 识别 | |||
| CAS号 | 106-46-7 | ||
| ChemSpider | 13866817 | ||
| SMILES |
| ||
| ChEBI | 28618 | ||
| RTECS | CZ4550000 | ||
| KEGG | C07092 | ||
| 性质 | |||
| 化学式 | C6H4Cl2 | ||
| 摩尔质量 | 146.992 g/mol g·mol⁻¹ | ||
| 密度 | 1.25 g/cm3, 固体 | ||
| 熔点 | 53.5 ℃ | ||
| 沸点 | 174 ℃ | ||
| 溶解性(水) | 10.5 mg/100 mL (20℃) | ||
| 危险性 | |||
| 警示术语 | R:R36, R40, R50/53 | ||
| 安全术语 | S:S2, S36/37, S46, S60, S61 | ||
| 欧盟分类 | Carc. Cat. 3 | ||
| NFPA 704 | |||
| 闪点 | 66 ℃ | ||
| 相关物质 | |||
| 相关化学品 | 邻二氯苯、间二氯苯 | ||
| 若非注明,所有数据均出自标准状态(25 ℃,100 kPa)下。 | |||
对二氯苯是苯的二个氢被氯原子取代后形成的化合物,分子式为C6H4Cl2,系统命名法写作1,4-二氯苯(英语:1,4-Dichlorobenzene),常用作樟脑丸。
性质
熔点53.5℃、沸点174℃。常温下会借由升华而散发出强烈气味,由于其强烈气味,尽管空气中只有微量的存在也能闻到气味。[1]
制取
对二氯苯通过苯的取代反应制得:
- C6H6 + 2 Cl2 → C6H4Cl2 + 2 HCl
反应中的主要杂质是1,2取代的异构体邻二氯苯,因为邻二氯苯和氯苯熔点远远低于室温,可利用对二氯苯相对较高的熔点53.5°C,通过分离结晶纯化。[1]
用途
对环境与人体健康的危害
对二氯苯难溶于水,不易被土壤生物分解,与许多芳香烃类相同,对二氯苯是脂溶性的,并会在脂肪组织中积累。
美国卫生和公共服务部(DHHS)和国际癌症研究机构(IARC)认为对二氯苯是一种致癌物质,虽然没有足够的证据。[4] 美国环境保护署(EPA)设立的最高标准为饮用水中不超过75微克/升。[5] 同时对二氯苯也是EPA登记的农药。
美国职业安全和健康管理局(OSHA)关于劳动安全设立的标准是对于每天8小时,每周工作40小时的人,工作场所空气中对二氯苯不得超过75ppm。
参考资料
- ^ 1.0 1.1 1.2 Rossberg, M.; Lendle, W.; Pfleiderer, G.; Tögel, A.; Dreher, E. L.; Langer, E.; Rassaerts, H.; Kleinschmidt, P.; Strack, H. Ullmann's Encyclopedia of Industrial Chemistry. 2006. ISBN 3527306730. doi:10.1002/14356007.a06_233.pub2.
|chapter=被忽略 (帮助) - ^ National Pesticide Information Center – Mothballs Case Profile (PDF). [10 August 2009]. (原始内容 (PDF)存档于2010年6月22日).
- ^ Fahey, D. R.; Ash, C. E. Mechanism of poly(p-phenylene sulfide) growth from p-dichlorobenzene and sodium sulfide. Macromolecules. 1991, 24 (15): 4242. doi:10.1021/ma00015a003.
- ^ Preamble to the IARC Monographs (页面存档备份,存于互联网档案馆) definition of "Group 2B: Possibly carcinogenic to humans", the International Agency for Research on Cancer classification of this chemical
- ^ Consumer Factsheet on: PARA-DICHLOROBENZENE (p-DCB). 28 November 2006 [10 August 2009]. (原始内容存档于2009年10月6日).
外部链接
- 国际化学品安全卡0037
- Mothball sniffing warning issued (页面存档备份,存于互联网档案馆), BBC News, 27 July 2006
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Text is available under the CC BY-SA 4.0 license; additional terms may apply.
Images, videos and audio are available under their respective licenses.