Ksanomelin
 | |
| IUPAC ime | |
|---|---|
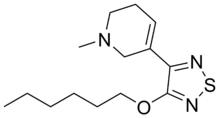
3-(4-heksoksi-1,2,5-tiadiazol-3-il)-1-metil-5,6-dihidro-2H-piridin | |
| Identifikatori | |
| CAS broj | 131986-45-3 |
| ATC kod | None |
| PubChem | CID 60809 |
| ChemSpider | 54797 |
| UNII | 9ORI6L73CJ |
| KEGG | D06330 |
| ChEMBL | CHEMBL21536 |
| Hemijski podaci | |
| Formula | C14H23N3OS |
| Molarna masa | 281,42 g/mol |
| |
Ksanomelin (LY-246,708, Lumeron, Memkor) je agonist muskarisnkog acetilholinskog receptora sa umerenom selektivnošću za M1 i M4 podtipove.[1][2][3][4] Nije poznato da ksanomelin deluje kao antagonist M5 receptora.[5] On je bio ispitivan za lečenje Alchajmerove bolesti i šizofrenije, posebno kognitivnih i negativnih simptoma.[6] Gastrointestinalne nuspojave su dovele do visoke stope napuštanja kliničkih ispitivanja.[7][8] Uprkos tome, za ksanomelin je pokazano da ima umerenu efikasnost u lečenju simptoma šizofrenije. Jedna nedavna studija je utvrdila robustno poboljšanje pri verbalnom učenju i u kratkotrajnoj memoriji usled tretmana ksanomelinom.[9]
Reference
[уреди | уреди извор]- ^ Farde L; Suhara T; Halldin C; et al. (1996). „PET study of the M1-agonists [11C]xanomeline and [11C]butylthio-TZTP in monkey and man”. Dementia (Basel, Switzerland). 7 (4): 187—95. PMID 8835881.
- ^ Jakubík J, Michal P, Machová E, Dolezal V (2008). „Importance and prospects for design of selective muscarinic agonists” (PDF). Physiological Research / Academia Scientiarum Bohemoslovaca. 57 Suppl 3: S39—47. PMID 18481916.
- ^ Woolley ML, Carter HJ, Gartlon JE, Watson JM, Dawson LA (2009). „Attenuation of amphetamine-induced activity by the non-selective muscarinic receptor agonist, xanomeline, is absent in muscarinic M4 receptor knockout mice and attenuated in muscarinic M1 receptor knockout mice”. European Journal of Pharmacology. 603 (1-3): 147—9. PMID 19111716. doi:10.1016/j.ejphar.2008.12.020.
- ^ Heinrich JN; Butera JA; Carrick T; et al. (2009). „Pharmacological comparison of muscarinic ligands: historical versus more recent muscarinic M1-preferring receptor agonists”. European Journal of Pharmacology. 605 (1-3): 53—6. PMID 19168056. doi:10.1016/j.ejphar.2008.12.044.
- ^ Grant MK, El-Fakahany EE (2005). „Persistent binding and functional antagonism by xanomeline at the muscarinic M5 receptor”. The Journal of Pharmacology and Experimental Therapeutics. 315 (1): 313—9. PMID 16002459. doi:10.1124/jpet.105.090134.
- ^ Lieberman JA, Javitch JA, Moore H (2008). „Cholinergic agonists as novel treatments for schizophrenia: the promise of rational drug development for psychiatry”. The American Journal of Psychiatry. 165 (8): 931—6. PMID 18676593. doi:10.1176/appi.ajp.2008.08050769.
- ^ Messer WS (2002). „The utility of muscarinic agonists in the treatment of Alzheimer's disease”. Journal of Molecular Neuroscience : MN. 19 (1-2): 187—93. PMID 12212779. doi:10.1007/s12031-002-0031-5.
- ^ Mirza NR, Peters D, Sparks RG (2003). „Xanomeline and the antipsychotic potential of muscarinic receptor subtype selective agonists”. CNS Drug Reviews. 9 (2): 159—86. PMID 12847557. doi:10.1111/j.1527-3458.2003.tb00247.x.
- ^ Shekhar A; Potter WZ; Lightfoot J; et al. (2008). „Selective muscarinic receptor agonist xanomeline as a novel treatment approach for schizophrenia”. The American Journal of Psychiatry. 165 (8): 1033—9. PMID 18593778. doi:10.1176/appi.ajp.2008.06091591.
Vidi još
[уреди | уреди извор]Spoljašnje veze
[уреди | уреди извор]Text is available under the CC BY-SA 4.0 license; additional terms may apply.
Images, videos and audio are available under their respective licenses.
