芳香环
芳香环是一类有机碳氢芳香化合物,简称芳环[1],概括了纯碳环与杂环两大类。若是成环的原子仅为碳,则特称为芳烃[2]。
芳香环拥有共轭的平面环体系,原子间成键并不是不连续的单双键交替,而是被离域π电子云覆盖。典型的芳香环化合物是苯和吲哚。
| 四元环 | 含四元环的稠环 | |
|---|---|---|
 苯并环丁烯 |
||
| 五元环 | 含五元环的稠环 | |
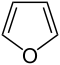 呋喃 |
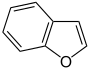 苯并呋喃 |
 异苯并呋喃 |
 吡咯 |
 吲哚 |
 异吲哚 |
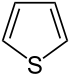 噻吩 |
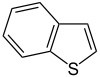 苯并[b]噻吩 |
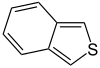 苯并[c]噻吩 |
 咪唑 |
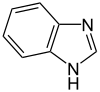 苯并咪唑 |
 嘌呤 |
 吡唑 |
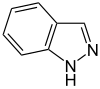 吲唑 |
|
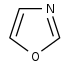 𫫇唑 |
 苯并𫫇唑 |
|
 异𫫇唑 |
 苯并异𫫇唑 |
|
 噻唑 |
 苯并噻唑 |
|
| 六元环 | 含六元环的稠环 | |
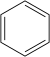 苯 |
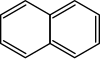 萘 |
 蒽 |
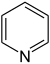 吡啶 |
 喹啉 |
 异喹啉 |
 吡嗪 |
 喹喔啉 |
 吖啶 |
 嘧啶 |
 喹唑啉 |
 菲 |
 哒嗪 |
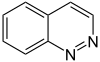 噌啉 |
|
分类
单环
简单的单环芳香化合物一般是五元环(如吡咯)或六元环(如吡啶),
多环与杂环
混合芳香环例如萘和嘌呤是简单环的合并,并共用它们连接的键。当环中有非碳元素的时候,简单的芳香环就变为了杂环。例如有氧、氮、硫。
含氮的芳香环
含氮的芳香环可以分为非碱性芳香环和碱性芳香环。
含氧和硫的芳香环
在有氧和硫的杂环中,杂环原子中的一对孤对电子参与形成离域π电子云(类似于非碱性含氮杂环),另外一对则延伸至环平面以外(类似于碱性含氮杂环),因此显碱性。
芳香性化合物的特性
- 分子必须构成环状
- 每一个原子必须有p轨域,并且每个p轨域必须是完全共轭
- 要么分子是平面的,要么满足同芳香性或莫比乌斯芳香性
- 分子必须有奇数对π电子,必须满足休克尔规则 (Hückel's rule):(4n+2)个π电子,n为自然数
与之对比的是,有4n个π电子的分子是反芳香性的
参考文献
参见
| |||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Text is available under the CC BY-SA 4.0 license; additional terms may apply.
Images, videos and audio are available under their respective licenses.
