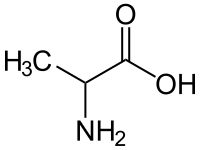
丙氨酸
| 丙氨酸 | |
|---|---|
| |

| |
| IUPAC名 Alanine 2-胺基丙酸 | |
| 别名 | 2-氨基丙酸 |
| 缩写 | Ala, A |
| 识别 | |
| CAS号 | 338-69-2(右旋) 56-41-7(左旋) 302-72-7(外消旋体) |
| PubChem | 5950 |
| ChemSpider | 64234 (右旋), 5735 (左旋), 582 (外消旋体) |
| SMILES |
|
| InChI |
|
| InChIKey | QNAYBMKLOCPYGJ-UHFFFAOYAA |
| ChEBI | 16977 |
| KEGG | C01401 |
| IUPHAR配体 | 720 |
| 性质 | |
| 化学式 | C3H7NO2 |
| 摩尔质量 | 89.09 g·mol−1 |
| 外观 | 白色粉末 |
| 密度 | 1.424 g/cm3 |
| 熔点 | 258 °C(531 K) |
| 溶解性(水) | 167.2 g/L (25 °C) |
| log P | -0.68[1] |
| pKa | pKa1 = 2.35(-COOH) pKa2 = 9.87(-NH3+)[2] |
| 磁化率 | -50.5·10−6 cm3/mol |
| 若非注明,所有数据均出自标准状态(25 ℃,100 kPa)下。 | |
丙氨酸(Alanine,简写为Ala或A)是一种氨基酸,于1850年由阿道夫·斯特雷克通过乙醛、氨和氢氰酸的反应首次合成。[3][4][5]它含有胺基和羧酸,二者都与中心碳原子相连,中心碳原子也带有甲基侧链。因此,它的IUPAC系统命名为2-胺基丙酸,并且它属于非极性氨基酸。
生物体内合成方式

化学合成方式
外消旋的丙氨酸可由乙醛、氯化铵和氰化钠进行斯特雷克氨基酸合成反应[6]合成。
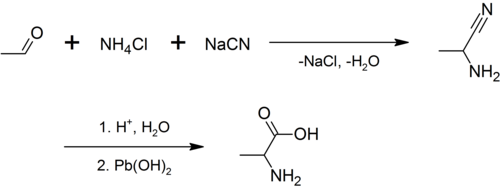
参考资料
- ^ L-alanine_msds. [2019-08-16]. (原始内容存档于2017-06-16).
- ^ Haynes, William M. (编). CRC化學和物理手冊 第97版. CRC Press. 2016: 5–88. ISBN 978-1498754286.
- ^ Strecker A. Ueber die künstliche Bildung der Milchsäure und einen neuen, dem Glycocoll homologen Körper [On the artificial formation of lactic acid and a new substance homologous to glycine]. Annalen der Chemie und Pharmacie. 1850, 75 (1): 27–45 [2023-04-24]. doi:10.1002/jlac.18500750103. (原始内容存档于2022-01-07) (德语). Strecker names alanine on p. 30.
- ^ Strecker A. Ueber einen neuen aus Aldehyd – Ammoniak und Blausäure entstehenden Körper [On a new substance arising from acetaldehyde–ammonia [i.e., 1-aminoethanol] and hydrocyanic acid]. Annalen der Chemie und Pharmacie. 1854, 91 (3): 349–351 [2023-04-24]. doi:10.1002/jlac.18540910309. (原始内容存档于2022-01-06) (德语).
- ^ Alanine. AminoAcidsGuide.com. 10 June 2018 [14 April 2019]. (原始内容存档于2022-01-06).
- ^ (1929) "dl-Alanine". Org. Synth. 9: 4; Coll. Vol. 1: 21.
|
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| ||||||||||||||||||||||||||||||||||||||||||||||
Text is available under the CC BY-SA 4.0 license; additional terms may apply.
Images, videos and audio are available under their respective licenses.

