Piridostigmin
Izvor: Wikipedija
| |||
| |||
| (IUPAC) ime | |||
|---|---|---|---|
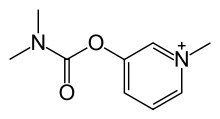
| 3-[(dimetilkarbamoil)oksi]-1-metilpiridinijum | |||
| Klinički podaci | |||
| Robne marke | Mestinon | ||
| AHFS/Drugs.com | Monografija | ||
| MedlinePlus | a682229 | ||
| Identifikatori | |||
| CAS broj | 155-97-5 | ||
| ATC kod | N07AA02 | ||
| PubChem[1][2] | 4991 | ||
| DrugBank | DB00545 | ||
| ChemSpider[3] | 4817 | ||
| UNII | 19QM69HH21 | ||
| KEGG[4] | D00487 | ||
| ChEMBL[5] | CHEMBL1115 | ||
| Hemijski podaci | |||
| Formula | C9H13N2O2 | ||
| Mol. masa | 181,212 g/mol | ||
| SMILES | eMolekuli & PubHem | ||
| |||
| Farmakokinetički podaci | |||
| Bioraspoloživost | 7,6 +/- 2,4% | ||
| Poluvreme eliminacije | 1,78 +/- 0,24 h | ||
| Izlučivanje | Renalno | ||
| Farmakoinformacioni podaci | |||
| Trudnoća | C(AU) C(US) | ||
| Pravni status | POM (UK) ℞-only (SAD) | ||
| Način primene | Oralno, intravenozno | ||
Piridostigmin je parasimpatomimetik i reverzibilni inhibitor holinesteraze. Pošto je ova suposanca kvaternarni amin, ona se slabo apsorbuje u stomaku i ne prelazi kroz krvno moždanu barijeru, izuzev u stresnim situacijama.[6]
Piridostigmin, 3-[(dimetilaminokarbonil)oksi]-1-metil piridinijum bromid, se može sintetisati iz 3-hidroksipiridina reakcijom sa dimetilaminokarbamoil hloridom, čime se formira 3-(dimetilaminokarbamoil)piridin. Daljom reakcijom sa metilbromidom nastaje piridostigmin.[7]
- ↑ Li Q, Cheng T, Wang Y, Bryant SH (2010). „PubChem as a public resource for drug discovery.”. Drug Discov Today 15 (23-24): 1052-7. DOI:10.1016/j.drudis.2010.10.003. PMID 20970519.
- ↑ Evan E. Bolton, Yanli Wang, Paul A. Thiessen, Stephen H. Bryant (2008). „Chapter 12 PubChem: Integrated Platform of Small Molecules and Biological Activities”. Annual Reports in Computational Chemistry 4: 217-241. DOI:10.1016/S1574-1400(08)00012-1.
- ↑ Hettne KM, Williams AJ, van Mulligen EM, Kleinjans J, Tkachenko V, Kors JA. (2010). „Automatic vs. manual curation of a multi-source chemical dictionary: the impact on text mining”. J Cheminform 2 (1): 3. DOI:10.1186/1758-2946-2-3. PMID 20331846.
- ↑ Joanne Wixon, Douglas Kell (2000). „Website Review: The Kyoto Encyclopedia of Genes and Genomes — KEGG”. Yeast 17 (1): 48–55. DOI:10.1002/(SICI)1097-0061(200004)17:1<48::AID-YEA2>3.0.CO;2-H.
- ↑ Gaulton A, Bellis LJ, Bento AP, Chambers J, Davies M, Hersey A, Light Y, McGlinchey S, Michalovich D, Al-Lazikani B, Overington JP. (2012). „ChEMBL: a large-scale bioactivity database for drug discovery”. Nucleic Acids Res 40 (Database issue): D1100-7. DOI:10.1093/nar/gkr777. PMID 21948594.
- ↑ Gulf War Syndrome: More Complex Than Middle East Politics. JWatch Psychiatry 1997;1997:15-15.
- ↑ R. Urban, U.S. Patent 2.572.579 (1951).
- Brenner, G. M. (2000). Pharmacology. Philadelphia, PA: W.B. Saunders Company. ISBN 0-7216-7757-6
- Canadian Pharmacists Association (2000). Compendium of Pharmaceuticals and Specialties (25th ed.). Toronto, ON: Webcom. ISBN 0-919115-76-4
- Neal, M.J. (2002). Medical Pharmacology at a Glance (5th ed.). London, England: Blackwell Publishing. ISBN 1-4051-3360-0
Text is available under the CC BY-SA 4.0 license; additional terms may apply.
Images, videos and audio are available under their respective licenses.

