For faster navigation, this Iframe is preloading the Wikiwand page for
偏二甲肼.
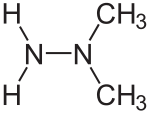
偏二甲肼,或称1,1-二甲基联氨、偏二甲基聯胺、偏二甲基肼,分子式(CH3)2NNH2,英文缩写UDMH(Unsymmetrical dimethylhydrazine),无色易燃液体。
二甲胺与亚硝酸作用后经还原而得。


二甲胺和氯胺反应而得。

- 高比冲液体火箭燃料:优点在于有高比冲值,与氧化剂接触即自动着火,利於火箭產品使用。

- 其中常规氧化剂为四氧化二氮:

- 做为液体火箭燃料,可在常温下保存和使用。这与需要低温的液氧-煤油的液体火箭燃料方案相比,具有更便捷的军事用途。所以苏联的R-36、SS-19、質子號火箭、中国的东风-5及其衍生的长征二号至长征四号火箭,都使用偏二甲肼-四氧化二氮常温方案。美國的泰坦系列運載火箭、三角洲系列運載火箭等則將偏二甲肼和聯氨一比一混合(稱航空肼50)後,再與四氧化二氮配合使用。
偏二甲肼是一种强还原剂,有毒,可致癌,且能透过皮肤直接进入人体内。故使用时需要作好防护措施
若偏二甲肼被释放到大气中,会成为N-亚硝基二甲胺,一种持久的致癌物与地下水污染物[1]
|
|---|
| | 无机肼 | |
|---|
| | 有机肼 | 脂肪肼/芳香肼 | |
|---|
| 酰肼(单取代) |
- 甲酰肼
- 4-PTSC
- 己二酰肼
- Adjudin
- 伞菌氨酸
- Benmoxin
- 卡屈嗪
- Carbazide
- 卡比多巴
- 碳酰肼
- Daminozide
- 双肼屈嗪
- 恩屈嗪
- 鹿花菌素
- HBT
- 肼屈嗪
- 异丙氯肼
- 异丙烟肼
- 异卡波肼
- 异烟肼
- 美巴那肼
- Metfendrazine
- 甲基肼
- Nialamide
- 奥他莫辛
- PEH
- 苯乙肼
- 苯异丙肼
- Phenoxypropazine
- Pildralazine
- Pimagedine
- 匹戊肼
- 甲苄肼
- 沙夫肼
- 氨基脲
- 缩氨脲
|
|---|
| 酰肼(二取代) | |
|---|
|
|---|
|
{{bottomLinkPreText}}
{{bottomLinkText}}
This page is based on a Wikipedia article written by
contributors (read/edit).
Text is available under the
CC BY-SA 4.0 license; additional terms may apply.
Images, videos and audio are available under their respective licenses.
{{current.index+1}} of {{items.length}}
Thanks for reporting this video!
This browser is not supported by Wikiwand :(
Wikiwand requires a browser with modern capabilities in order to provide you with the best reading experience.
Please download and use one of the following browsers:
An extension you use may be preventing Wikiwand articles from loading properly.
If you're using HTTPS Everywhere or you're unable to access any article on Wikiwand, please consider switching to HTTPS (https://www.wikiwand.com).
An extension you use may be preventing Wikiwand articles from loading properly.
If you are using an Ad-Blocker, it might have mistakenly blocked our content.
You will need to temporarily disable your Ad-blocker to view this page.
✕
This article was just edited, click to reload
Please click Add in the dialog above
Please click Allow in the top-left corner,
then click Install Now in the dialog
Please click Open in the download dialog,
then click Install
Please click the "Downloads" icon in the Safari toolbar, open the first download in the list,
then click Install
{{::$root.activation.text}}
Follow Us
Don't forget to rate us
Oh no, there's been an error
Please help us solve this error by emailing us at
support@wikiwand.com
Let us know what you've done that caused this error, what browser you're using, and whether you have any special extensions/add-ons installed.
Thank you!