N-(1-萘基)乙二胺
| N-(1-萘基)乙二胺[1] | |
|---|---|

| |

| |
| IUPAC名 N1-(Naphthalen-1-yl)ethane-1,2-diamine N1-(1-萘基)乙基-1,2-二胺 | |
| 别名 | 萘乙二胺 |
| 识别 | |
| CAS号 | 551-09-7 1465-25-4(二鹽酸鹽) |
| PubChem | 15107 |
| ChemSpider | 14379 |
| SMILES |
|
| InChI |
|
| InChIKey | NULAJYZBOLVQPQ-UHFFFAOYAJ |
| Beilstein | 2803527 |
| ChEBI | 53510 |
| 性质 | |
| 化学式 | C12H14N2 |
| 摩尔质量 | 186.25 g·mol−1 |
| 外观 | 灰白色結晶 |
| 氣味 | 無味 |
| 密度 | 380 kg/m3 |
| 熔点 | 189 °C(462 K)([2]) |
| 溶解性(水) | 30 g/1000 mL(二鹽酸鹽,20 °C) |
| 危险性 | |
GHS危险性符号
| |
| GHS提示词 | Warning |
| H-术语 | H315, H319 |
| P-术语 | P264, P280, P302+352, P305+351+338, P332+313, P337+313 |
| 主要危害 | 刺激性 |
| NFPA 704 | |
| 闪点 | 非易燃 |
| 若非注明,所有数据均出自标准状态(25 ℃,100 kPa)下。 | |
N-(1-萘基)乙二胺是一種有機化合物,化学式为C12H14N2。它作為格里斯试剂的一部分出售,可用於格里斯試驗對血液中的硝酸鹽、亞硝酸鹽和磺胺進行定量無機分析。
生產
[编辑]N-(1-萘基)乙二胺可由1-萘胺與2-氯乙胺反應製備,或通过1-溴萘和乙二胺反应生成,产物溶于盐酸后重结晶可制得。[3]其通常以二鹽酸鹽形式出售。
性質
[编辑]N-(1-萘基)乙二胺會發生大多數萘胺和伯胺的典型反應,例如重氮化反應。與其類似物乙二胺類似,它也可以作為雙齒配體提供多種錯位化合物。然而,它是一種較弱的雙齒配體,因為萘胺基團中的氮原子由於共振引起的電荷分散而弱配位。例如在水溶液中與氯亞鉑酸鉀反應生成[N-(1-萘基)乙二胺]二氯鉑(II)。[4]
它也可以和酰氯反应,生成相应的酰胺。[5]
用途
[编辑]N-(1-萘基)乙二胺二鹽酸鹽廣泛用於比色法定量分析水樣中的硝酸鹽和亞硝酸鹽。它很容易在亞硝酸鹽存在下發生重氮偶聯反應,生成顏色較深的偶氮化合物。含有亞硝酸根離子的樣品首先被中和,然後在0-5 °C下用稀鹽酸處理得到亞硝酸。然後加入過量但固定體積的磺胺和N-(1-萘基)乙二胺二鹽酸鹽溶液。以亞硝酸為限制試劑,偶氮偶聯反應生成偶氮染料,相對於亞硝酸根離子定量:
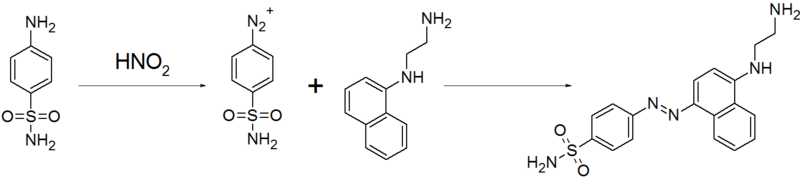
形成的重氮化合物產生了陽性結果的典型紅色。然後用色度計測量所得溶液的顏色強度,並根據校準曲線檢查以確定亞硝酸根離子濃度。[6]
要定量分析硝酸鹽濃度,可先將樣品通過銅鎘柱,將硝酸根離子定量還原為亞硝酸根離子,但應針對原始樣品中存在的亞硝酸根離子調整結果。[7]
這種方法可以應用於許多方面,包括確定污水或其他生物樣品(如細胞內液)中的硝酸鹽/亞硝酸鹽濃度,前提是樣品必須為透明無色的。
在類似的情況下,可以使用相同原理檢測血液中的磺胺水平。
參考資料
[编辑]- ^ Chemicals and reagents, 2008-2010, Merck
- ^ Wagner, J.; Prusova, J. Preparation of N-(1-naphthyl)ethylenediamine. Casopis Lekaru Ceskych, 1951. 90: 20-22. ISSN: 0008-7335.
- ^ 赵德章, 邹政红, 何金. 盐酸萘乙二胺的制备方法. 2011. CN102173996A.
- ^ Horst Kunkely, Arnd Vogler. Synthesis and Electronic Spectra of (N-1-Naphthyl-ethylenediamine)-dichloroplatinum(II). Fluorescence of the Appended Naphthyl Substituent. Z. Naturforsch. 2002, 57b: 709–711.
- ^ Seema Bag, Rekha Tulsan, Abha Sood, Hyejin Cho, Hana Redjeb, Weihong Zhou, Harry LeVine, Béla Török, Marianna Török. Sulfonamides as multifunctional agents for Alzheimer’s disease. Bioorganic & Medicinal Chemistry Letters. 2015-02, 25 (3): 626–630 [2021-03-24]. doi:10.1016/j.bmcl.2014.12.006. (原始内容存档于2020-02-28) (英语).
- ^ G. H. Jerffery; J. Bassett; J. Mendham; R. C. Denney. Colorimetry and Spectrophotometry. Vogel's Textbook of Quantitative Chemical Analysis, 5th Edition. Longman. 1989: 702. ISBN 0-582-44693-7.
- ^ [1] (页面存档备份,存于互联网档案馆), Nitrate analysis protocol and principles.
Text is available under the CC BY-SA 4.0 license; additional terms may apply.
Images, videos and audio are available under their respective licenses.

