Skattebøl rearrangement
The Skattebøl rearrangement is an organic reaction for converting a geminal dihalo cyclopropane to an allene using an organolithium base.[1][2] This rearrangement reaction is named after its discoverer, Lars Skattebøl, Professor emeritus at the University of Oslo. It proceeds through a carbene reaction intermediate:

When the cyclopropane ring is fitted with a 2-vinyl group, a cyclopentadiene is formed through a so-called foiled carbene intermediate.[3][4] This process is more generally known as a vinylcyclopropane rearrangement.
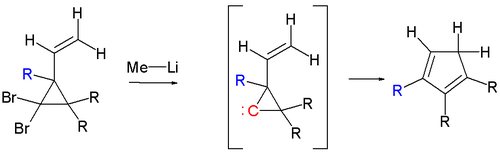
The reaction is closely related to the earlier Doering-LaFlamme procedure (Doering-LaFlamme allene synthesis), in which a gem-dibromocyclopropane is treated with an alkali metal to form the same cyclopropylidene intermediate.
Text is available under the CC BY-SA 4.0 license; additional terms may apply.
Images, videos and audio are available under their respective licenses.
