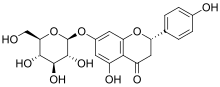
Prunin
| |
| |
| Names | |
|---|---|
| IUPAC name
(2S)-7-(β-D-Glucopyranosyloxy)-4′,5-dihydroxyflavan-4-one
| |
| Systematic IUPAC name
(2S)-5-Hydroxy-2-(4-hydroxyphenyl)-7-{[(2S,3R,4S,5S,6R)-3,4,5-trihydroxy-6-(hydroxymethyl)oxan-2-yl]oxy}-2,3-dihydro-4H-1-benzopyran-4-one | |
| Other names
Naringenin-7-O-glucoside
| |
| Identifiers | |
3D model (JSmol)
|
|
| ChEBI | |
| ChemSpider | |
| ECHA InfoCard | 100.007.696 |
| KEGG | |
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C21H22O10 | |
| Molar mass | 434.397 g·mol−1 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Prunin is a flavanone glycoside found in immature citrus fruits[1][2] and in tomatoes.[3] Its aglycone form is called naringenin.
Metabolism
Glucosidase breaks prunin into glucose and naringenin.
Text is available under the CC BY-SA 4.0 license; additional terms may apply.
Images, videos and audio are available under their respective licenses.
