Paternò–Büchi reaction
| Paternò–Büchi reaction | |
|---|---|
| Named after | Emanuele Paternò George Büchi |
| Reaction type | Ring forming reaction |
| Identifiers | |
| Organic Chemistry Portal | paterno-buechi-reaction |
| RSC ontology ID | RXNO:0000083 |
The Paternò–Büchi reaction, named after Emanuele Paternò and George Büchi, who established its basic utility and form,[1][2] is a photochemical reaction, specifically a 2+2 photocycloaddition, which forms four-membered oxetane rings from an excited carbonyl and reacting with an alkene.[3]

Here an electronically excited carbonyl group is added to a ground state olefin yielding an oxetane.
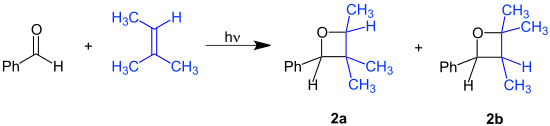
With substrates benzaldehyde and 2-methyl-2-butene the reaction product is a mixture of structural isomers:
Another substrate set is benzaldehyde and furan[4] or heteroaromatic ketones and fluorinated alkenes.[5]
The alternative strategy for the above reaction is called the Transposed Paternò−Büchi reaction.
Text is available under the CC BY-SA 4.0 license; additional terms may apply.
Images, videos and audio are available under their respective licenses.