PAK2
Serine/threonine-protein kinase PAK 2 is an enzyme that in humans is encoded by the PAK2 gene.[5][6]
PAK2 is one of three members of Group I PAK family of serine/threonine kinases.[7][8] The PAKs are evolutionary conserved.[9] PAK2 and its cleaved fragment localize in both the cytoplasmic or nuclear compartments. PAK2 signaling modulates apoptosis,[10] endothelial lumen formation,[11] viral pathogenesis,[12] and cancer including, breast,[13] hepatocarcinoma,[14] and gastric [15] and cancer, at-large.[16]
Discovery
The human PAK2 was identified as a downstream effector of Rac or Cdc42.[7][8]
Gene and spliced variants
The PAK2 gene is about 92.7-kb long. The gene contains 15 exons and generates three alternatively spliced transcripts - two of which code proteins of 524 amino acids and 221 amino acids, while the third one is a 371-bp non-coding RNA transcript(Gene from review) There are two transcripts generated from the murine PAK2 gene, a 5.7-kb transcript coding a 524 amino acids long polypeptide and a 1.2-kb long non-coding RNA transcript.
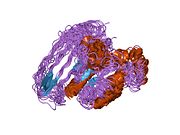
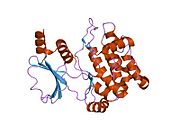
Protein domains
Similar to PAK1, PAK2 contains a p21-binding domain (PBD) and an auto-inhibitory domain (AID) and exists in an inactive conformation.[16]
The p21 activated kinases (PAK) are critical effectors that link Rho GTPases to cytoskeleton reorganization and nuclear signaling. The PAK proteins are a family of serine/threonine kinases that serve as targets for the small GTP binding proteins, CDC42 and RAC1, and have been implicated in a wide range of biological activities. The protein encoded by this gene is activated by proteolytic cleavage during caspase-mediated apoptosis, and may play a role in regulating the apoptotic events in the dying cell.[17]
Function
The p21 activated kinases (PAK) are critical effectors that link Rho GTPases to cytoskeleton reorganization and nuclear signaling. The PAK proteins are a family of serine/threonine kinases that serve as targets for the small GTP binding proteins, CDC42 and RAC1, and have been implicated in a wide range of biological activities. The protein encoded by this gene is activated by proteolytic cleavage during caspase-mediated apoptosis, and may play a role in regulating the apoptotic events in the dying cell.[18]
Upstream activators
PAK2 kinase activity is stimulated by transforming growth factor β in fibroblasts,[19] by proteinase inhibitor alpha2-macroglobulin binding to GRP78 in prostate cancer cells,[20] by its phosphorylation by AMP-activated protein kinase in stem and cancer cells [21] and eryptosis.[22] PAK2 is cleaved through activated caspase-3 in fibroblast and cancer cells exposed to ultraviolet,[23] hyperosmotic shock,[24] and ionizing radiation.[25]
Inhibitors
The levels of PAK2 activation in experimental systems are inhibited by synthetic PAK-inhibitors and miRs. For example, FRAX1036 differentially inhibits PAK2 and PAK1 activities;[26] FRAX597 suppresses PAK2 activity in neurofibromatosis type 2 (NF2)-associated tumorigenesis;[27] and miR-23b and miR-137 inhibits PAK2 expression in tumor cells.[28][29] Insulin stimulation of neuronal cells also antagonizes PAK2 kinase activity, leading to an increased glucose uptake.[30]
Downstream targets
PAK2-mediated phosphorylation of merlin at S518 modulates its tumor suppressor activity,[31] c-Jun phosphorylation at T2, T8, T89, T93 and T286 contributes to the growth of growth factor-stimulated melanoma cells,[32] Caspase-7 phosphorylation at S30, T173 and S239 inhibits apoptotic activity in breast cancer cells,[13] Paxillin phosphorylation at S272 and S274 activates ADAM10 protease,[33] and STAT5 phosphorylation at S779 modulates BCL-ABL-mediated leukemogenesis.[34] PAK2 activity negatively regulates the function and expression of c-Myc: PAK2 phosphorylation of c-Myc at T358-S373-T400 inhibits its transactivation function [35] and PAK2 depletion stimulates c-Myc expression during granulocyte-monocyte lineage.[36]
Text is available under the CC BY-SA 4.0 license; additional terms may apply.
Images, videos and audio are available under their respective licenses.