Manganese stearate
| |
| Names | |
|---|---|
| Other names
Manganese(II) stearate, manganese distearate, manganese(2+) dioctadecanoate
| |
| Identifiers | |
3D model (JSmol)
|
|
| ChemSpider | |
| ECHA InfoCard | 100.020.110 |
| EC Number |
|
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C 36H 70MnO 4 | |
| Molar mass | 621.89 |
| Appearance | Pale pink powder |
| Density | g/cm3 |
| Boiling point | 359.4 °C (678.9 °F; 632.5 K) |
| insoluble | |
| Hazards | |
| GHS labelling: | |
| Warning | |
| H302, H312, H315, H319, H332, H335 | |
| Flash point | 162.4 °C (324.3 °F; 435.5 K) |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
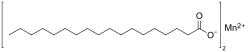
Manganese stearate is a metal-organic compound, a salt of manganese and stearic acid with the chemical formula C
36H
70MnO
4.[1][2] The compound is classified as a metallic soap, i.e. a metal derivative of a fatty acid.[3]
Synthesis
[edit]Manganese stearate is synthesized by the reaction of stearic acid with sodium hydroxide, followed by reacting with manganese chloride.[4]
Also, the reaction of manganese(II) acetate with stearic acid.[5]
Physical properties
[edit]The compound forms pale pink powder.[6]
Insoluble in water.[6]
Uses
[edit]The compound is used in organic synthesis reactions.[6]
Also as an oxidant additive for oxo-biodegradable polymers (for example, high-density polyethylene).[7]
References
[edit]- ^ "Manganese Stearate". American Elements. Retrieved 6 March 2023.
- ^ "NCATS Inxight Drugs — MANGANESE STEARATE". National Center for Advancing Translational Sciences. Retrieved 6 March 2023.
- ^ "CAS 3353-05-7 Manganese Stearate - Alfa Chemistry". alfa-chemistry.com. Retrieved 6 March 2023.
- ^ Aras, Neny Rasnyanti M.; Arcana, I Made (2015). "Synthesis of manganese stearate for high density polyethylene (HDPE) and its biodegradation". AIP Conference Proceedings. 1677 (1): 070024. Bibcode:2015AIPC.1677g0024A. doi:10.1063/1.4930728. Retrieved 6 March 2023.
- ^ "manganese stearate". chemsrc.com. Retrieved 6 March 2023.
- ^ a b c "Manganese Stearate | CAS 3353-05-7". Santa Cruz Biotechnology. Retrieved 6 March 2023.
- ^ Roy, Prasun Kumar; Singh, Priyanka; Kumar, Devendra; Rajagopal, Chitra (2010). "Manganese stearate initiated photo-oxidative and thermo-oxidative degradation of LDPE, LLDPE and their blends". Journal of Applied Polymer Science. 117: 524–533. doi:10.1002/app.31252.
| Manganese(-I) | |
|---|---|
| Manganese(0) | |
| Manganese(I) | |
| Manganese(II) | |
| Manganese(II,III) | |
| Manganese(II,IV) |
|
| Manganese(III) | |
| Manganese(IV) | |
| Manganese(V) | |
| Manganese(VI) | |
| Manganese(VII) | |
Salts and covalent derivatives of the stearate ion | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Text is available under the CC BY-SA 4.0 license; additional terms may apply.
Images, videos and audio are available under their respective licenses.
