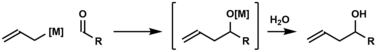
Carbonyl allylation
In organic chemistry, carbonyl allylation describes methods for adding an allyl anion to an aldehyde or ketone to produce a homoallylic alcohol.[1] The carbonyl allylation was first reported in 1876 by Alexander Zaitsev and employed an allylzinc reagent.[2]
Enantioselective versions
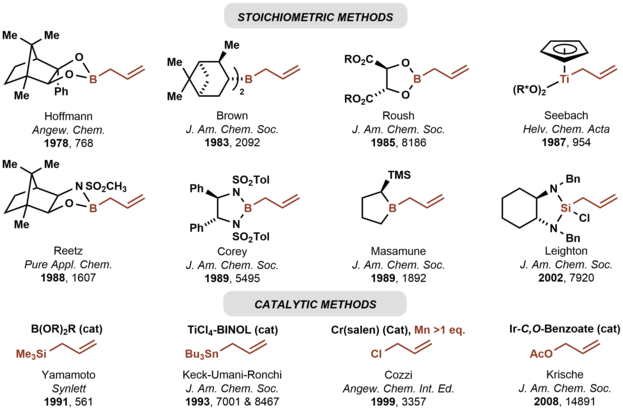
In 1978, Hoffmann reported the first asymmetric carbonyl allylation using a chiral allylmetal reagent, an allylborane derived from camphor.[3][4] Such methods utilize preformed allyl metal reagents. The approach is well developed using allyl boranes[5]
(13)

As illustrated by the Keck allylation,[6] catalytic enantioselective additions of achiral allylmetal reagents to carbonyl compounds also are possible by organostannane additions.[7]
Allylic boronate and -borane reagents have also been developed for enantioselective addition to carbonyls—in this class of reactions, the allylic boron reagent confers stereochemical control[5]
(13)

Catalysis
In 1991, Yamamoto disclosed the first catalytic enantioselective method for carbonyl allylation, which employed a chiral boron Lewis acid-catalyst in combination with allyltrimethylsilane.[8] Numerous other catalytic enantioselective methods for carbonyl allylation followed.[9][6] Catalytic variants of the Nozaki-Hiyama-Kishi reaction represent an alternative method for asymmetric carbonyl allylation, but stoichiometric metallic reductants are required.[10]
Whereas the aforementioned asymmetric carbonyl allylations rely on preformed allylmetal reagents, the Krische allylation exploits allyl acetate for enantioselective carbonyl allylation.[11] Selected methods for asymmetric carbonyl allylation are summarized below.
Use in total synthesis
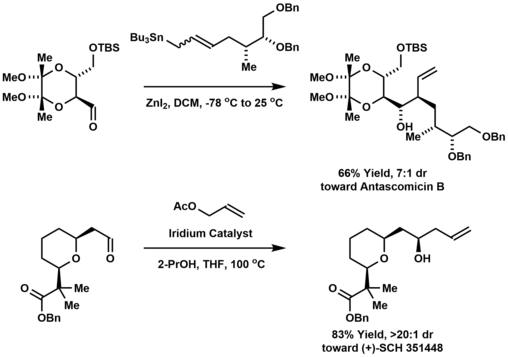
Carbonyl allylation has been employed in the synthesis of polyketide natural products and other oxygenated molecules with a contiguous array of stereocenters. For example, allylstannanation of a threose-derived aldehyde affords the macrolide antascomicin B, which structurally resembles FK506 and rapamycin, and is a potent binder of FKBP12.[12] The Krische allylation was used to prepare the polyketide (+)-SCH 351448, a macrodiolide ionophore bearing 14 stereogenic centers.[13]
Older primary literature
- Brown, Herbert C.; Jadhav, Prabhakar K. (April 1983). "Asymmetric carbon-carbon bond formation via .beta.-allyldiisopinocampheylborane. Simple synthesis of secondary homoallylic alcohols with excellent enantiomeric purities". Journal of the American Chemical Society. 105 (7): 2092–2093. doi:10.1021/ja00345a085. ISSN 0002-7863.
- Hayashi, Tamio; Konishi, Mitsuo; Kumada, Makoto (1982-09-01). "Optically active allylsilanes. 2. High stereoselectivity in asymmetric reaction with aldehydes producing homoallylic alcohols". Journal of the American Chemical Society. 104 (18): 4963–4965. doi:10.1021/ja00382a046.
- Roush, William R.; Walts, Alan E.; Hoong, Lee K. (1985-12-01). "Diastereo- and enantioselective aldehyde addition reactions of 2-allyl-1,3,2-dioxaborolane-4,5-dicarboxylic esters, a useful class of tartrate ester modified allylboronates". Journal of the American Chemical Society. 107 (26): 8186–8190. doi:10.1021/ja00312a062.
- Brown, Herbert C.; Jadhav, Prabhakar K. (April 1983). "Asymmetric carbon-carbon bond formation via .beta.-allyldiisopinocampheylborane. Simple synthesis of secondary homoallylic alcohols with excellent enantiomeric purities". Journal of the American Chemical Society. 105 (7): 2092–2093. doi:10.1021/ja00345a085. ISSN 0002-7863.
- Kinnaird, James W. A.; Ng, Pui Yee; Kubota, Katsumi; Wang, Xiaolun; Leighton, James L. (2002-07-01). "Strained Silacycles in Organic Synthesis: A New Reagent for the Enantioselective Allylation of Aldehydes". Journal of the American Chemical Society. 124 (27): 7920–7921. doi:10.1021/ja0264908. ISSN 0002-7863. PMID 12095334.
- Short, Robert P.; Masamune, Satoru (March 1989). "Asymmetric allylboration with B-allyl-2-(trimethylsilyl)borolane". Journal of the American Chemical Society. 111 (5): 1892–1894. doi:10.1021/ja00187a061. ISSN 0002-7863.
- Corey, E. J.; Yu, Chan Mo; Kim, Sung Soo (July 1989). "A practical and efficient method for enantioselective allylation of aldehydes". Journal of the American Chemical Society. 111 (14): 5495–5496. doi:10.1021/ja00196a082. ISSN 0002-7863.
Text is available under the CC BY-SA 4.0 license; additional terms may apply.
Images, videos and audio are available under their respective licenses.
