Aflatoxin total synthesis
Aflatoxin total synthesis concerns the total synthesis of a group of organic compounds called aflatoxins. These compounds occur naturally in several fungi. As with other chemical compound targets in organic chemistry, the organic synthesis of aflatoxins serves various purposes. Traditionally it served to prove the structure of a complex biocompound in addition to evidence obtained from spectroscopy. It also demonstrates new concepts in organic chemistry (reagents, reaction types) and opens the way to molecular derivatives not found in nature. And for practical purposes, a synthetic biocompound is a commercial alternative to isolating the compound from natural resources. Aflatoxins in particular add another dimension because it is suspected that they have been mass-produced in the past from biological sources as part of a biological weapons program.[1][2]
The synthesis of racemic aflatoxin B1 has been reported by Buechi et al. in 1967[3] and that of racemic aflatoxin B2 by Roberts et al. in 1968[4] The group of Barry Trost of Stanford University is responsible for the enantioselective total synthesis of (+)-Aflatoxin B1 and B2a in 2003.[5] In 2005 the group of E. J. Corey of Harvard University presented the enantioselective synthesis of Aflatoxin B2.[6]
Aflatoxin B2 synthesis
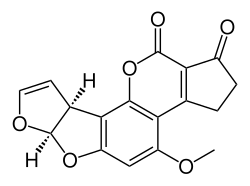
[edit]The total synthesis of Aflatoxin B2 is a multistep sequence that begins with a [2+3]cycloaddition between the quinone 1 and the 2,3-Dihydrofuran. This reaction is catalyzed by a CBS catalyst and is enantioselective. The next step is the orthoformylation of reaction product 2 in a Duff reaction. The hydroxyl group in 3 is esterified with triflic anhydride which adds a triflate protecting group. This step enables a Grignard reaction of the aldehyde group in 4 with methylmagnesiumbromide to the alcohol 5 which is then oxidized with the Dess-Martin periodinane to the ketone 6. A Baeyer-Villiger oxidation converts the ketone to an ester (7) and a reduction with Raney nickel converts the ester into an alcohol and removes the triflic acid group. In the final step the coumarin skeleton is added to 9 by a combined coupling reaction with zinc carbonate of the vinyl bromide in 8 and a transesterification step between the phenol group and the ethyl ester group.
 |
| Aflatoxin B2 total synthesis |
|---|
References
[edit]- ^ Zilinskas, Raymond A. (1997). "Iraq's Biological Weapons. The Past as Future?". JAMA. 278 (5): 418–424. doi:10.1001/jama.1997.03550050080037.
- ^ Venkataramana, M.; Chandranayaka, S.; Prakash, H. S.; Niranjana, S. R. (2015). "Mycotoxins Relevant to Biowarfare and Their Detection". In Gopalakrishnakone, P.; Balali-Mood, Mahdi; Llewellyn, Lyndon; Singh, Bal Ram (eds.). Biological Toxins and Bioterrorism. Springer. pp. 295–319. ISBN 978-94-007-5869-8.
- ^ Buechi, George; Foulkes, D. M.; Kurono, Masayasu; Mitchell, Gary F.; Schneider, Richard Stephen (1967). "The total synthesis of racemic aflatoxin B1". Journal of the American Chemical Society. 89 (25): 6745–53. doi:10.1021/ja01001a062. PMID 6063661.
- ^ Roberts, John C.; Sheppard, A. H.; Knight, J. A.; Roffey, Patrick (1968). "Studies in mycological chemistry. Part XXII. Total synthesis of (±)-aflatoxin-B2". Journal of the Chemical Society C: Organic. 1: 22–24. doi:10.1039/J39680000022. PMID 5688875.
- ^ Trost, B. M.; Toste, F. D. "Palladium Catalyzed Kinetic and Dynamic Kinetic Asymmetric Transformations of γ-Acyloxybutenolides. Enantioselective Total Synthesis of (+)-Aflatoxin B1 and B2a". J. Am. Chem. Soc. 2003, 125, 3090–3100. doi:10.1021/ja020988s
- ^ Zhou, G.; Corey, E. J. "Short, Enantioselective Total Synthesis of Aflatoxin B2 Using an Asymmetric [3+2]-Cycloaddition Step". J. Am. Chem. Soc. 2005, 127, 11958–11959. doi:10.1021/ja054503m
Text is available under the CC BY-SA 4.0 license; additional terms may apply.
Images, videos and audio are available under their respective licenses.
