For faster navigation, this Iframe is preloading the Wikiwand page for
Plantilya:Infobox plutonium .
Plutonium
94
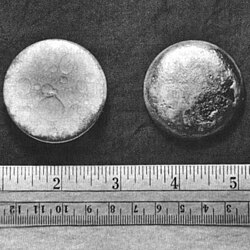
Panagway
silvery white, tarnishing to dark gray in air
Kinatibuk-ang mga kinaiya
Ngalan, simbolo, kaiphan
plutonium, Pu, 94
Paglitok
[[Help:Pronunciation respelling key|ploo-Plantilya:Smallcaps all -nee-əm ]]
Kategoriyang elemento
aktinido
Group, period, block
[[group element|]], 7, f
Gibug-aton sa atomo
(244)
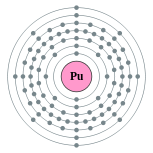
Kontorno sa elektron
[Rn ] 5f6 7s2 Electron shells of plutonium (2, 8, 18, 32, 24, 8, 2)
History
Pagkadiskobre
Glenn T. Seaborg, Arthur Wahl, Joseph W. Kennedy, Edwin McMillan (1940–1)
Physical properties
Phase
magahi
Density (near r.t.)
19.816 g·cm−3
Liquid density at m.p.
16.63 g·cm−3
Melting point
912.5 K , 639.4 °C, 1182.9 °F
Boiling point
3505 K, 3228 °C, 5842 °F
Heat of fusion
2.82 kJ·mol−1
Heat of vaporization
333.5 kJ·mol−1
Molar heat capacity
35.5 J·mol−1 ·K−1
Vapor pressure
P (Pa)
1
10
100
1 k
10 k
100 k
at T (K)
1756
1953
2198
2511
2926
3499
Atomic properties
Oxidation states
8, 7, 6, 5, 4 , 3, 2, 1
Electronegativity
1.28 (Pauling scale)
Ionization energies
1st: 584.7 kJ·mol−1
Atomic radius
159 pm
Covalent radius
187±1 pm
Miscellanea
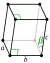
Crystal structure
monoclinic
Magnetic ordering
paramagnetic[1]
Electrical resistivity
(0 °C) 1.460 µΩ·m
Thermal conductivity
6.74 W·m−1 ·K−1
Thermal expansion
(25 °C) 46.7 µm·m−1 ·K−1
Speed of sound
2260 m·s−1
Young's modulus
96 GPa
Shear modulus
43 GPa
Poisson ratio
0.21
CAS registry number
7440-07-5
Most stable isotopes
Main article: Isotopes of plutonium
iso
NA
half-life
DM
DE (MeV)
DP
238 Pu
trace
87.74 y
SF
204.66[2]
α
5.5
234 U
239 Pu
100%
2.41 × 104 y
SF
207.06
α
5.157
235 U
240 Pu
trace
6.5 × 103 y
SF
205.66
α
5.256
236 U
241 Pu
syn
14 y
β−
0.02078
241 Am
SF
210.83
242 Pu
trace
3.73 × 105 y
SF
209.47
α
4.984
238 U
244 Pu
trace
8.08 × 107 y
α
4.666
240 U
SF
· r
Plantilya:Template reference list
↑ Magnetic susceptibility of the elements and inorganic compounds , in Lide, D. R., ed. (2005). CRC Handbook of Chemistry and Physics (86th ed.). Boca Raton (FL): CRC Press. ISBN 0-8493-0486-5 ↑ Magurno, B.A.; Pearlstein, S. (eds.) Workshop on nuclear data evaluation methods and procedures , Upton, NY, USA, 22 Septmber 1980, vol. II (1981), pp. 835 ff
{{bottomLinkPreText}}
{{bottomLinkText}}
This page is based on a Wikipedia article written by
contributors (read /edit ).CC BY-SA 4.0 license; additional terms may apply.
Plantilya:Infobox plutonium
{{current.index+1}} of {{items.length}}
Thanks for reporting this video!
This browser is not supported by Wikiwand :(
An extension you use may be preventing Wikiwand articles from loading properly.HTTPS Everywhere or you're unable to access any article on Wikiwand, please consider switching to HTTPS (https ://www.wikiwand.com).
An extension you use may be preventing Wikiwand articles from loading properly.Ad-Blocker , it might have mistakenly blocked our content.
You will need to temporarily disable your Ad-blocker to view this page.
✕
This article was just edited, click to reload
Please click Add in the dialog above
Please click Allow in the top-left corner, Install Now in the dialog
Please click Open in the download dialog, Install
Please click the "Downloads" icon in the Safari toolbar, open the first download in the list, Install
{{::$root.activation.text}}
Follow Us
Don't forget to rate us
Oh no, there's been an error
Please help us solve this error by emailing us at
support@wikiwand.com
Let us know what you've done that caused this error, what browser you're using, and whether you have any special extensions/add-ons installed.
Thank you!